Highlight
Potential Gene Transfer Adjuvants to Improve Intrathecal Microencapsulated Interleukin-10 Gene Delivery
Achievement/Results
Ellen Dengler, an INCBN IGERT Trainee, is working on the application of silica nanoparticles as a non-viral platform to deliver gene transfer adjuvant molecules to the spinal cord, as a means to enhance therapeutic transgene uptake by resident cells surrounding the spinal cord, with the goal of achieving a relief of enduring neuropathic pain. This is an interdisciplinary effort involving neurobiologists and chemical and material engineers.
Pro-inflammatory signaling from spinal glia (astrocytes and microglia) is critical for pathological pain states (light touch sensitivity; allodynia). Interleukin-10 (IL-10) is an anti-inflammatory cytokine that suppresses the production and action of pro-inflammatory mediators. Previously, the group of Dr. Erin Milligan, Ellen Dengler’s advisor, has shown that two peri-spinal injections spaced discretely apart (5 hr. – 3 days) of a free plasmid DNA (125 µg total) encoding the IL-10 gene (pDNA-IL-10) reduces pro-inflammation and reverses neuropathic pain for up to 90 days in rats with chronic constriction injury (CCI) of the sciatic nerve. Within this “sensitization period”, phagocytic macrophage cell enrichment in the perispinal subarachnoid space was observed. Peri-spinal pDNA-IL-10 injections given outside the sensitization period failed to produce long duration pain relief. While encapsulating pDNA-IL-10 in poly-lactic-co-glycolic acid (PLGA) microparticles revealed similar pain reversal with a single injection at more than 10-fold lower doses, 1 mg PLGA was required to deliver the transgene, a PLGA dose that is potentially limiting for spinal clinical applications. While PLGA microparticles have demonstrated exceptional clinical safety for use in other applications, this dosage formulation is an example of a limiting characteristic of PLGA microparticles, such that these microparticles have a low drug-loading capacity.
The goal of this project is to take advantage of factors that activate peri-spinal macrophages to increase therapeutic IL-10 gene-containing phagosomes, while maintaining low therapeutic transgene & PLGA microparticle doses. Factors known to stimulate macrophages may act as “gene transfer” adjuvants, while also enhancing anti-inflammatory signaling during the sensitization period for subsequent enduring pain relief.
Address Goals
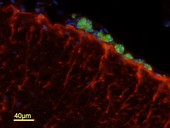
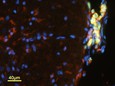
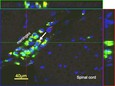
Recently, Ellen Dengler’s work has focused on determining the best candidate gene transfer adjuvant (GT-Adjvt) from several candidate compounds. dexamethasone (DEX) was chosen, because it is well characterized to act on macrophages and suppress proinflammatory processes. A second adjuvant was chosen, oligodeoxynucleotide (ODN) sequences, known to induce a rapid pro-inflammatory cytokine response. However, a delayed, anti-inflammatory IL-10 response follows, suggesting that ODNs may also stimulate macrophage phagocytosis and anti-inflammatory actions. A third adjuvant, mannose (MAN), activates scavenger receptors on macrophages that leads to both increased phagocytosis and anti-inflammatory activity. Initially, peri-spinal free GT-Adjvt was given, followed 3 days later by peri-spinal free pDNA-IL-10 at a dose previously reported as ineffective for pain relief. CCI neuropathic rats were assessed for reversal of allodynia for 90 days after injection. Subsequently, the best candidate GT-Adjvt was delivered by a novel synthetic nanoparticle, comprised of a porous silica core surrounded by a 1,2-dioleoyl-3-trimethylammonium-propane: cholesterol (DOTAP: Chol) lipid bilayer, termed a protocell. These protocells were found to be safe and non-toxic when injected intrathecally in rats. Protocells were observed to associate with perispinal macrophages, but not with astrocytes, and caused no overt adverse effects to the cells of the spinal cord (Figure 1).
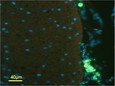
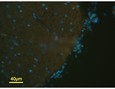
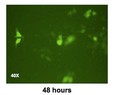
Protocells have numerous advantages over PLGA, including a large cargo capacity for small drugs, like GT-Adjvts. By modifying the silica core surface chemistry, protocells can be tuned to release cargo in synchrony with different cellular events in the meninges, and the lipid coating can be bioconjugated to small ligands for receptor-mediated cell targeting in the spinal cord. Ellen Dengler has demonstrated that pDNA-IL-10 encoding a reporter green fluorescent protein (GFP) remains functional following procedures required to adsorb pDNA-IL-10-GFP to protocells. Cell culture experiments using human embryonic kidney cells, which do not normally make IL-10 protein, resulted in expressed GFP and produced quantifiable levels of IL-10 protein following a 24-hour incubation with protocells loaded with pDNA-IL-10GFP (Figure 2). An earlier study revealed that ODNs loaded in protocells co-injected with pDNAIL-10-PLGA caused ~2wk pain reversal.
The work towards establishing the best gene transfer adjuvant is ongoing, and it remains to be determined which candidate adjuvant will best facilitate Il-10 gene transfer and enduring pain relief from peripheral neuropathy. The goal is to use a single injection, containing GT-Advts loaded on protocells and co-injected with a low dose of pDNA-IL-10 encapsulated in PLGA microparticles. Enhancing transgene uptake by the novel use of protocells containing transfer adjuvants and delivered to the central nervous system may facilitate not only gene therapies for pain relief, but will extend to neurodegenerative diseases.