Highlight
Increasing Efficacy for Lung Cancer Chemotherapeutics: Targeted Drug Delivery via the Protocell
Achievement/Results
According to current statistics reported by the Center for Disease Control and Prevention (CDC), lung cancer is the second most common cancer in both men and women. Lung cancer is also the cause of a third of all cancer related deaths in the United States. Astonishingly, more people die of lung cancer than of colon, breast, and prostate cancers combined. There is a definite necessity for advancement in the area of lung cancer research. Lung cancer is divided into two different types, including small cells and non-small cells. Since Non-Small Cell Lung Cancer (NSCLC) accounts for 85-90% of all lung cancer cases, the research project of Annikka L. Jensen, an INCBN IGERT Trainee, is focused on NSCLC. The typical survival for patients with advanced stage NSCLC is 6 months for untreated patients and only a mere 3 months more for patients treated with chemotherapy. The reported 5-year survival rate of patients with advanced NSCLC is less than 10%. These statistics strongly indicate a need to break through the ceiling of low clinical efficacy in lung cancer chemotherapeutics. Thus, increasing the clinical efficacy of lung cancer chemotherapeutics is an area of high interest, which will foster research that will have far reaching implications for increasing the quality of life for people suffering from lung cancer.
The therapeutic efficacy of traditionally delivered drugs is often hindered by non-specific toxicity that arises from the cytotoxic effects of the drug(s) on healthy cells. In efforts to improve therapeutic efficacy of cancer therapies, formulation methods of drug encapsulation or complexation with liposomes, polymers, or proteins have been employed. However, while treatments using such strategies are clinically available, they rely on passive target principles, such as the drug permeability and retention effect. Currently, there are few clinically-approved treatments that selectively target diseased cells via specific biomolecular interactions. The research project of Annikka Jensen is focusing on targeted drug delivery to cancer cells via engineered mesoporous nanoparticle-supported lipid bilayers (protocells), a new class of targeted nanocarriers developed at the University of New Mexico. This new class of targeted nanocarriers holds significant promise for improving the efficacy of cancer treatments, as their combination of specificity, stability, and enhanced cargo capacity allows for the capability of delivering a high concentration of the drug directly to the human cancer cell with minimal systemic toxicity. Initial in vitro experiments in the laboratories of Annikka Jensen’s advisor Prof. Jeffrey Brinker and her co-advisor Prof. Larry Sklar have demonstrated SP94-conjugated protocells were able to specifically target and kill human hepatocellular carcinoma (HCC) cells with orders of magnitude greater efficiency than ‘free’ or liposomal encapsulated drug, even in multiple drug resistant variants.
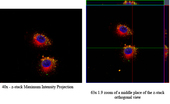
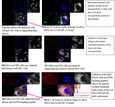
Increasing efficacy of non-small cell lung cancer chemotherapeutics via the EGFR-targeted protocell in vitro It has been shown previously that cells can actively sense nanoparticle shape via non covalent electrostatic and hydrogen bonding interactions. Certain shapes defined by aspect ratios stimulated filapodia formation and macropinocytosis. The following experiments were carried out to establish that shielding the surface silanols with a perfectly conformal coating that preserved shape but altered hydrogen bonding and surface charge would alter the internalization pathway of the mesoporous silica nanoparticles (MSNPs). To obtain the physiochemical characterization of the MSNP cores, transmission electron microscopy (TEM) experiments were carried out to determine the MSNP aspect ratio and pore structure, with shape and morphology of the MSNPs characterized by scanning electron microscopy (SEM). Spherical MSNPs had the aspect ratio of 1.0-1.2, diameter of ~110 nm, and uniform pore size of 2.5 nm. Rod cylindrical MSNPs had the aspect ratio of 2.1-2.5, with dimensions of 160-190/60-90 nm, and uniform pore size of 2.5 nm. Then, zeta potential experiments were performed to obtain the particle charge at a concentration of ~125 µgrams of protocells/ml in 0.5X PBS. Initial confocal fluorescence microscopy experiments were performed with the positive control, A549 cell line, and the negative controls, BEAS-2B cell line and MRC9 cell line, to establish that the non-small cell lung cancer cell lines will be able to have preferential delivery of the biologically active molecules based on the use of a targeting peptide. For these initial experiments, an established GE11 peptide, with sequence YHWYGYTPQNVI, was chosen as a targeting peptide as it binds to the highly over-expressed epidermal growth factor receptor (EGFR) in NSCLC cell lines.
The results of these initial binding studies showed that NSCLC cells could be selectively targeted using protocells bio-functionalized with EGFR targeting peptide, with no non-specific targeting to non-cancerous cells observed. After validating the EGFR targeting peptide for specific binding and internalization of the protocells in the A549 in vitro model, fluorescent plate reading techniques were used to determine the apparent binding affinities (Kd) for the EGFR targeting peptide at high (~2048 peptides), medium (~120 peptides), and low (6 peptides) densities on protocells (with varying aspect ratios and dimensions) using MSNP cores. Binding affinities of EGFR-targeting peptide on the protocells towards the A549 and MRC9 cells were also measured using flow cytometry. Co-localization experiments were performed to see if the EGFR-targeted protocells were being endocytosed via the EGFR mediated route into the early endosomes. For this portion of the experiments, immunohistochemistry with confocal microscopy and the Zen 2009 Light Edition software were used to determine the co-localization of the EGFR-targeted protocells.
Address Goals
Primary: Protocells have demonstrated remarkable cell targeting specificity and cancer cytotoxicity with hepatocarcinoma in vitro studies and show promise in serving as a universal targeted drug delivery system; however, their efficacy with non-small cell lung cancer in vitro remained unknown at the o0nset of this study. Results from Annikka Jensen’s research will contribute to world-class knowledge regarding the toxicity and therapeutic efficacy of this engineered nanoparticle drug delivery system in vitro, facilitating development of protocells for in vivo inhalation studies. In vivo inhalation biodistribution, toxicity, and pharmacokinetic studies will build the nation’s research capability by supporting the translation of protocells from the lab bench to the clinic to serve as a direct targeted drug delivery system for non-small cell lung cancer treatment.
Secondary: Although research and discovery are the primary goals of this project, building the Nation’s research capability is an important byproduct. Extending this research to in vitro NSCLC models will assist in leading to significant increases in the efficacy of current lung cancer chemotherapeutics by reducing the growth of human lung adenocarcinoma/carcinoma cells, including multiple drug resistant variants, to a greater degree than by using the corresponding ‘free’ or liposomal-delivered drugs. This will be achieved through harnessing the combined power of the protocell targeted drug delivery system and localized direct delivery of the chemotherapeutics via the inhalation route to the lungs. This discovery work will facilitate establishing the nation as a global leader in advancing the instrumental tools available to allow for revolutionizing the field of targeted drug delivery.